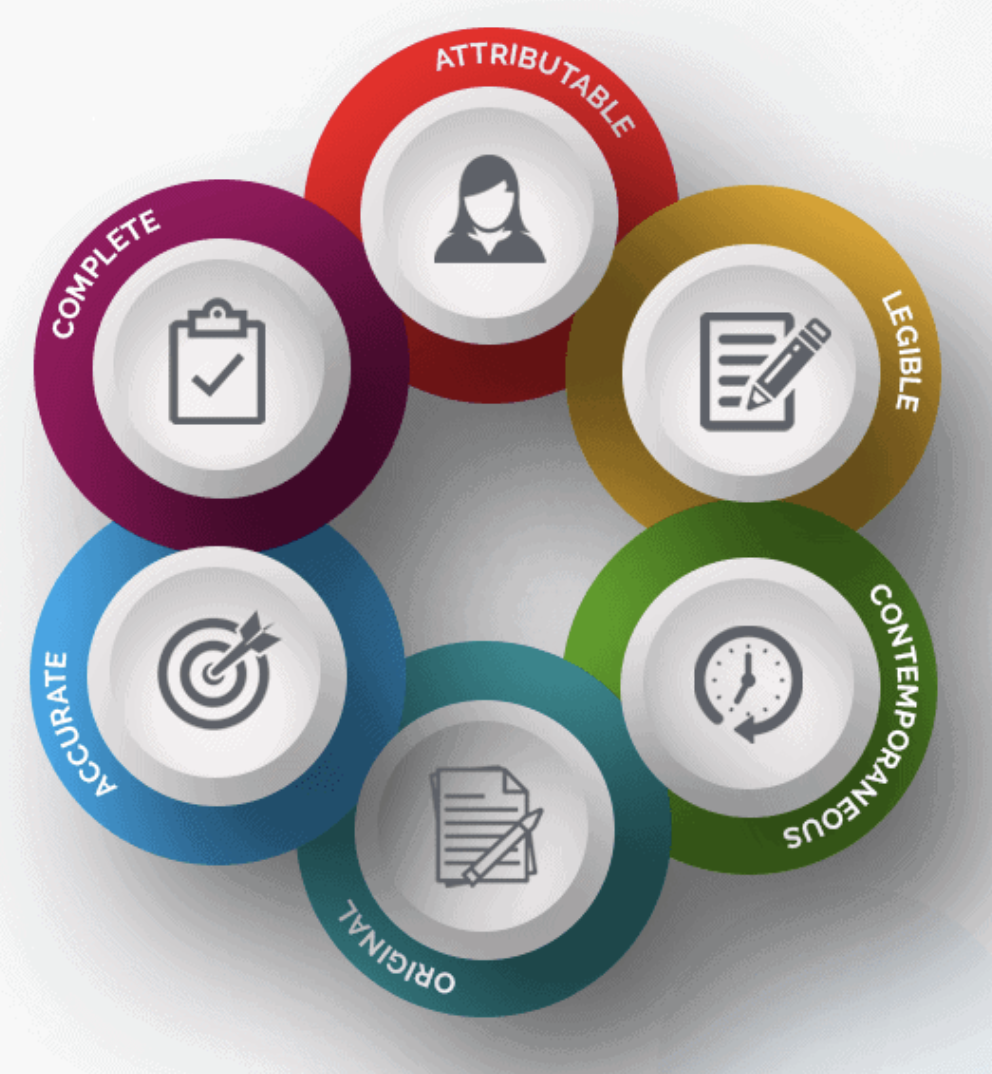
Products CTMS Lite
Our customized web-based application for the CRO’s helps to manage & execute clinical trials and streamline business operations.

Industry Transformation
Healthcare Provider Rethink Traditional Strategies
Design Strategies
Cloud technology allows Vertis Global CTMS Lite to offer the likewise strength to every size of the organization—from top 10 Pharma to startups and CROs—with no IT expertise required to configure Vertis Global CTMS Lite to the unique requirements of your study.
Agile development methodology fuels regular functional enhancements to Vertis Global CTMS Lite and the cloud puts those enhancements quickly into your research study. Thus, Vertis Global CTMS Lite always keeps pace with your clinical research processes.
Don’t battle expensive integrations or fragmented data. Vertis Global CTMS’s open architecture seamlessly exchanges information with other systems—pre-populating data, triggering events and eliminating duplicate work.
Our customizable dashboards allow clinical trial leaders to have a clear picture of clinical trials and make business informed decisions.
- Deep Industry expertise
- Analytics-led approach
- Agile Methodology
- Global Presence
- World-class strategic & technology partners
- Transparent business processes

